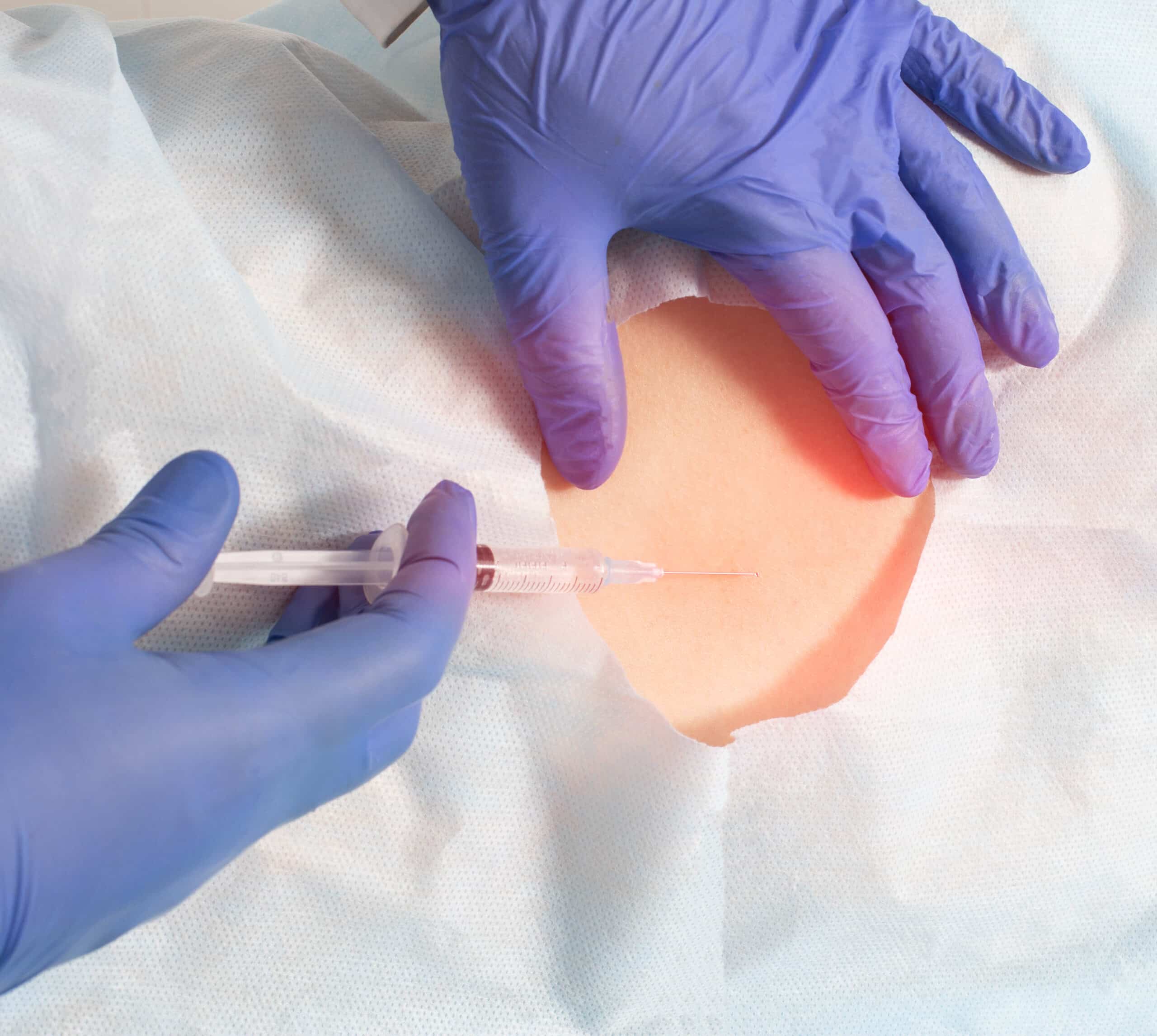
Injections
Injections of medications to decrease pain and inflammation have long been used in pain management. Traditionally, it has always been a combination of a steroid and a local anesthetic agent. The steroid acts to interrupt the inflammatory cascade that leads to pain and swelling. The local anesthetic provides immediate pain relief to provide diagnostic confirmation of the pain generator as well as to break the pain cycle in certain chronic pain syndromes.
Types of Injections
Living Life Again
At VSI, we celebrate every patient’s unique journey as a series of victories, regardless of how big or small. Our logo and brand were crafted for you, our incredible patients. Your progress towards your goals deserves the spotlight, reflecting our commitment to individualized treatment and care. Your Victories are what make VSI who we are! #MyVictoryStory
Discover the Benefits of Injections
- Decreases swelling and inflammation
- Reduces nerve irritation
- Pain relief
- May prevent operative treatment